Basic process design of Glatt powder synthesis for the production of homogeneous micro- and nanopowders with definable particle properties
Glatt Powder Synthesis in a Pulsating Gas Flow
How It Works
Glatt powder synthesis, based on a pulsating gas flow, is a process for the production, modification or coating of powder particles in the nano and micrometer range. The process takes place in a continuously operated vertical reaction tube.
Glatt Powder Synthesis in a pulsating gas stream – The principle process flow
The raw materials are sprayed into the tempered gas stream pulsating in the reactor chamber. Depending on the objective, the materials are dried, coated or thermally treated. This is followed by their rapid cooling and separation in the filter. The entire manufacturing process typically takes less than one second.
Schematic of the individual process steps for Glatt powder synthesis to produce homogeneous micro- and nanopowders with definable particle properties
Glatt Powder Synthesis in a pulsating gas flow – The process in detail
At first, a pulsating gas stream is generated at a temperature corresponding to the requirements. The raw material – a solution, suspension or solid is sprayed into the reactor chamber through a nozzle. Within the reactor, these materials meet the pulsating gas stream. Solutions and suspensions are atomized during spraying and break up even further due to pulsation and thermal shock. Due to the high specific surface area of these superfine droplets, the solvent evaporates very quickly. The originally dissolved material is dried to a very fine powder. The accelerated particle formation and phase transformation provides the desired reaction states and – depending on the starting product – enables the formation of unique structures. In this way, fine homogeneous particles are created from solutions, whereas suspensions allow the generation of core-shell particles. With the optional reactor heater, the particles can additionally be calcined at gas temperatures up to 1300°C in the same process step.
The thermal treatment defines the chemical, crystallographic and surface properties. A quencher, using a cold gas, quenches and “freezes” these structures in a fraction of a second. Finally, the material is deposited and isolated in various filters.
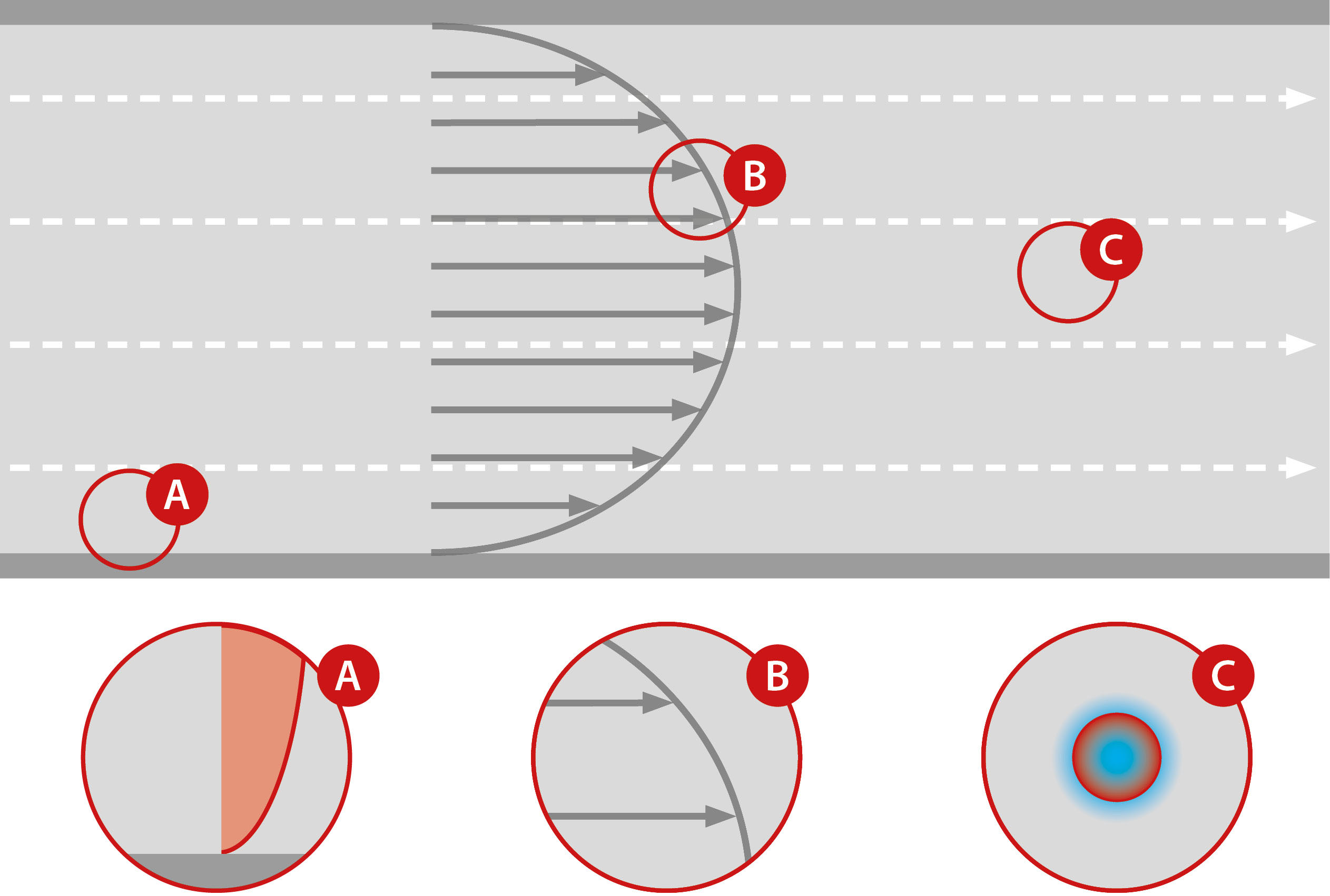
Glatt Powder Synthesis – Pulsation makes the difference
A pulsating gas flow has distinct advantages over a laminar gas flow. In a constant and laminar flow, the velocity vectors are aligned in parallel. Therefore, strong temperature and velocity gradients are formed perpendicual to the flow direction. As a result, powder particles – depending on their distance to the reactor axis – face very different process temperatures and residence times. In addition, the constant flow velocity just accelerates the particles until there is no longer any relative velocity to the process gas. Distinct boundary layers form around the particles, which hinder heat and mass transfer.
Disadvantages of non-pulsed gas flows:
(A) Temperature gradients result in different heat treatment depending on the position.
(B) Velocity gradients result in different residence times of the particles.
(C) Low relative velocity between gas stream and particles lead to boundary layers. This results in reduced mass and heat transfer.
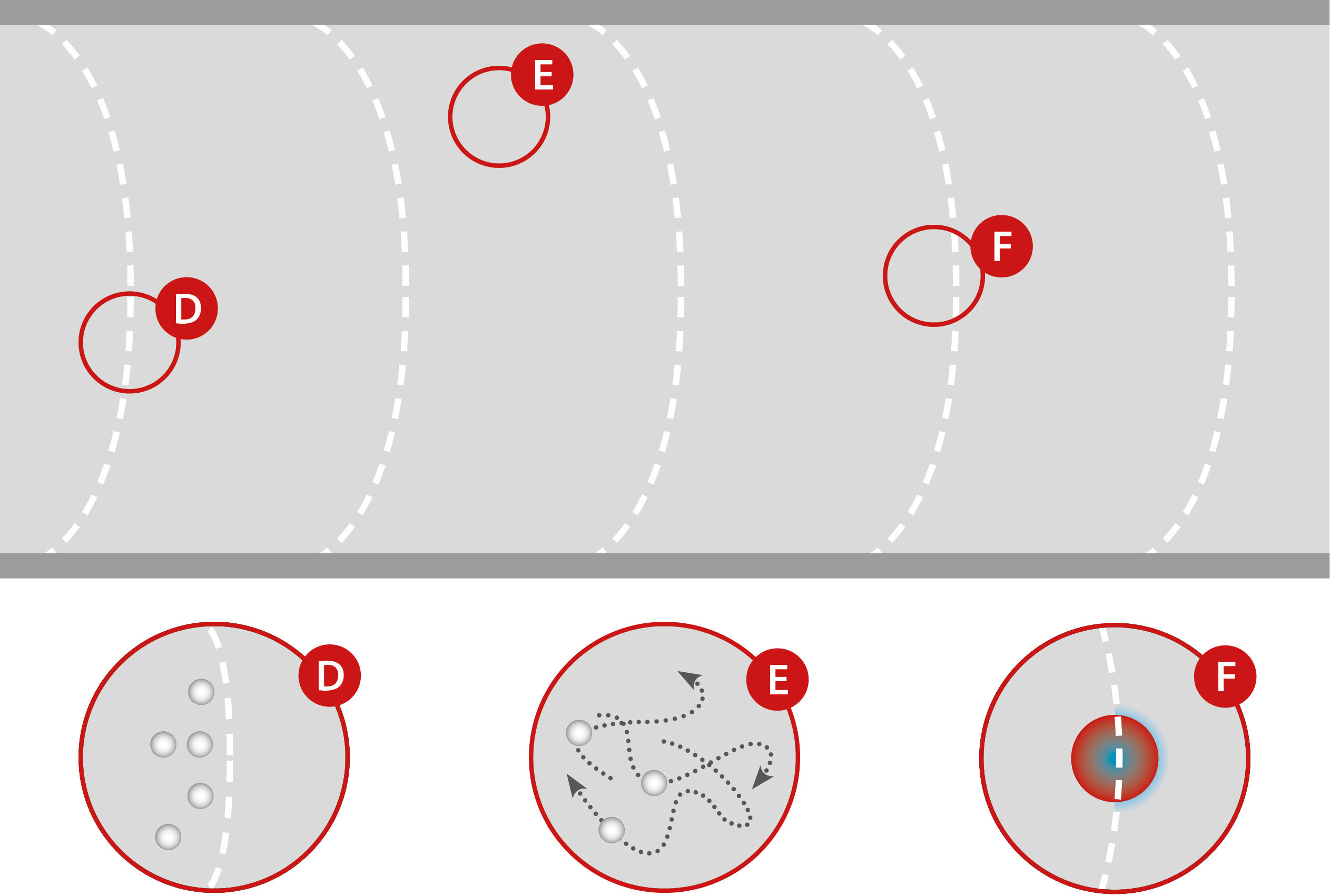
Advantages of pulsating gas flows:
(D) The impingement of the pressure waves produces superfine droplets by secondary atomization.
(E) Pulsation creates turbulence, which balances temperature and velocity in the gas stream while mixing the particles. This homogenizes the residence time.
(F) Impact of the pressure waves and the continuous relative velocity between particles and gas cause the boundary layers to break up. This results in improved mass and heat transfer.
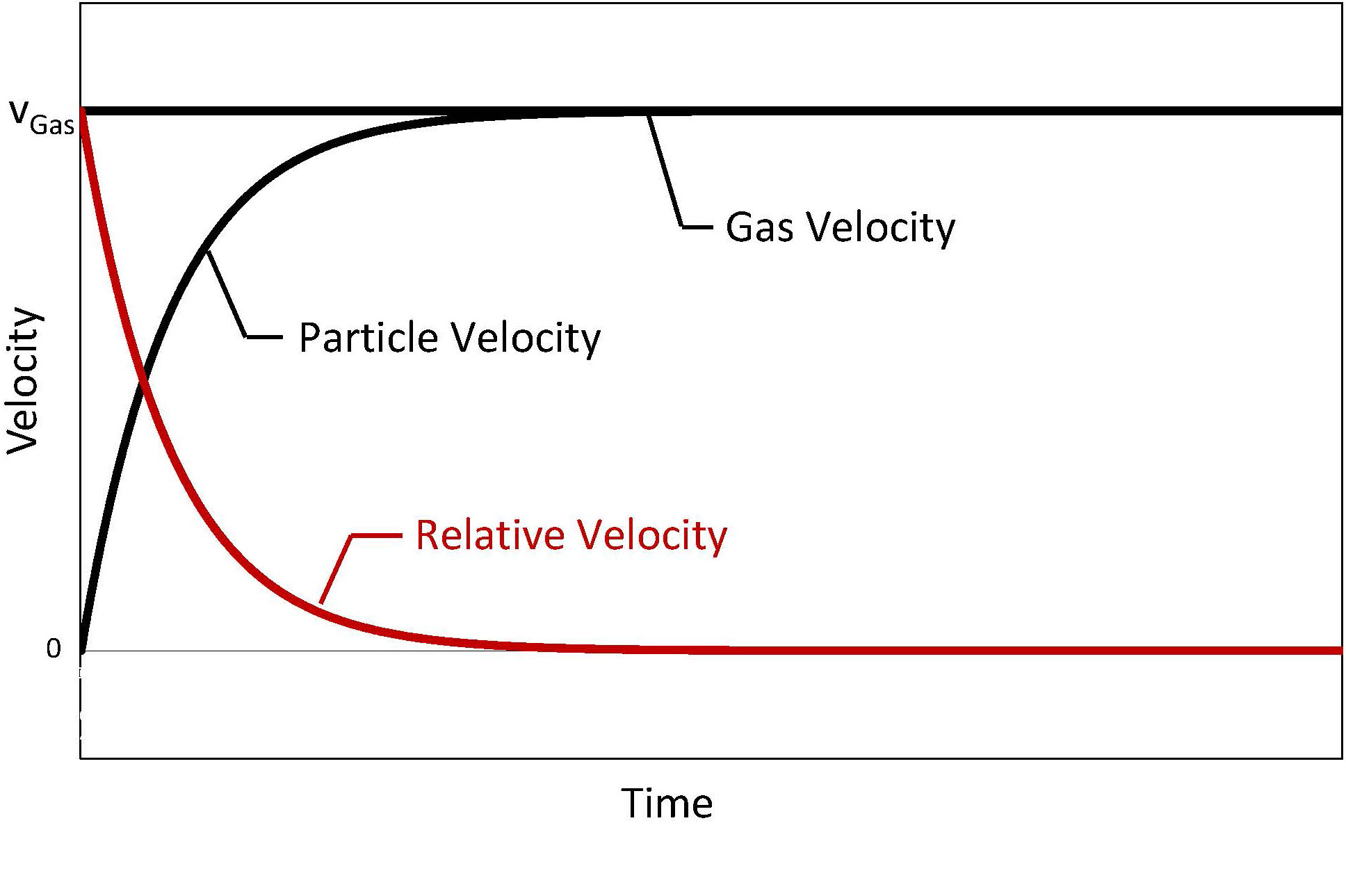
Pulsation in the gas flow of Glatt powder synthesis generates a highly turbulent gas flow. The associated exchange of gas and particles, even across the reactor, leads to homogenization of the gas temperature and balances residence times of the particles. In addition, the continuously increasing and decreasing flow velocity – in conjunction with the inertia of the particles – results in a relative velocity that persists over the entire length of the reactor. Boundary layers around the particles can only form very weakly and are additionally ablated by the impinging pressure waves. The result is an extremely high heat and mass transfer.
Velocity characteristics within non-pulsed gas streams
Velocity characteristics within pulsed gas streams
Further information on this topic and related topics can also be found in the following publications:
June 2021: ‘Fraunhofer IKTS operates world’s first flameless high-temperature powder synthesis plant’
Published article: ‘Ceramic Raw Materials from the Pulsating Hot Gas Stream’ PDF, English
Published article: ‘New Battery Material by Powder Synthesis’ PDF, English