Warum Auftragsfertigung kritisch und entscheidend ist (Fachbeitrag auf Englisch)
Jahrzehntelang haben große Unternehmen Produktionskapazitäten ausgelagert und damit die Globalisierung vorangetrieben – aber auch zum Niedergang der heimischen Industrie beigetragen. Durch die Digitalisierung macht es jedoch für immer mehr namhafte Global Player Sinn, die Produktion zurück nach Europa zu verlagern. Das Beispiel der Auftragsfertigung zeigt, wohin die Reise gehen kann. (Fachbeitrag in englischer Sprache)
- Autor: Dr. Michael Jacob ist Leiter des Bereiches Verfahrenstechnik bei Glatt Ingenieurtechnik, Deutschland. Er promovierte zum Thema „Experimentelle Untersuchungen und Modellierung von Prozessen in horizontalen Wirbelschichten zur Sprühgranulation“. Er ist außerdem Leiter des Glatt Technologiezentrums für Food, Feed & Fine Chemicals in Weimar.
- im Original veröffentlicht in der Fachzeitschrift ‚Agro FOOD Industry Hi Tech‘, Ausgabe 04.2019, TEKNOSCIENZE Srl
- tks | publisher, event organiser, media agency | Why contract manufacturing is both critical and crucial – tks | publisher, event organiser, media agency (teknoscienze.com)
Weitere Informationen zu diesem Thema und verwandten Themen finden Sie auch in den folgenden Veröffentlichungen:
‚Outside-In-Prozess vom Anlagenbauer: Von der Idee zur Marktreife – Zukunft wird im Team gemacht‘, Statement Gudrun Ding, Glatt Ingenieurtechnik GmbH, PDF, deutsch
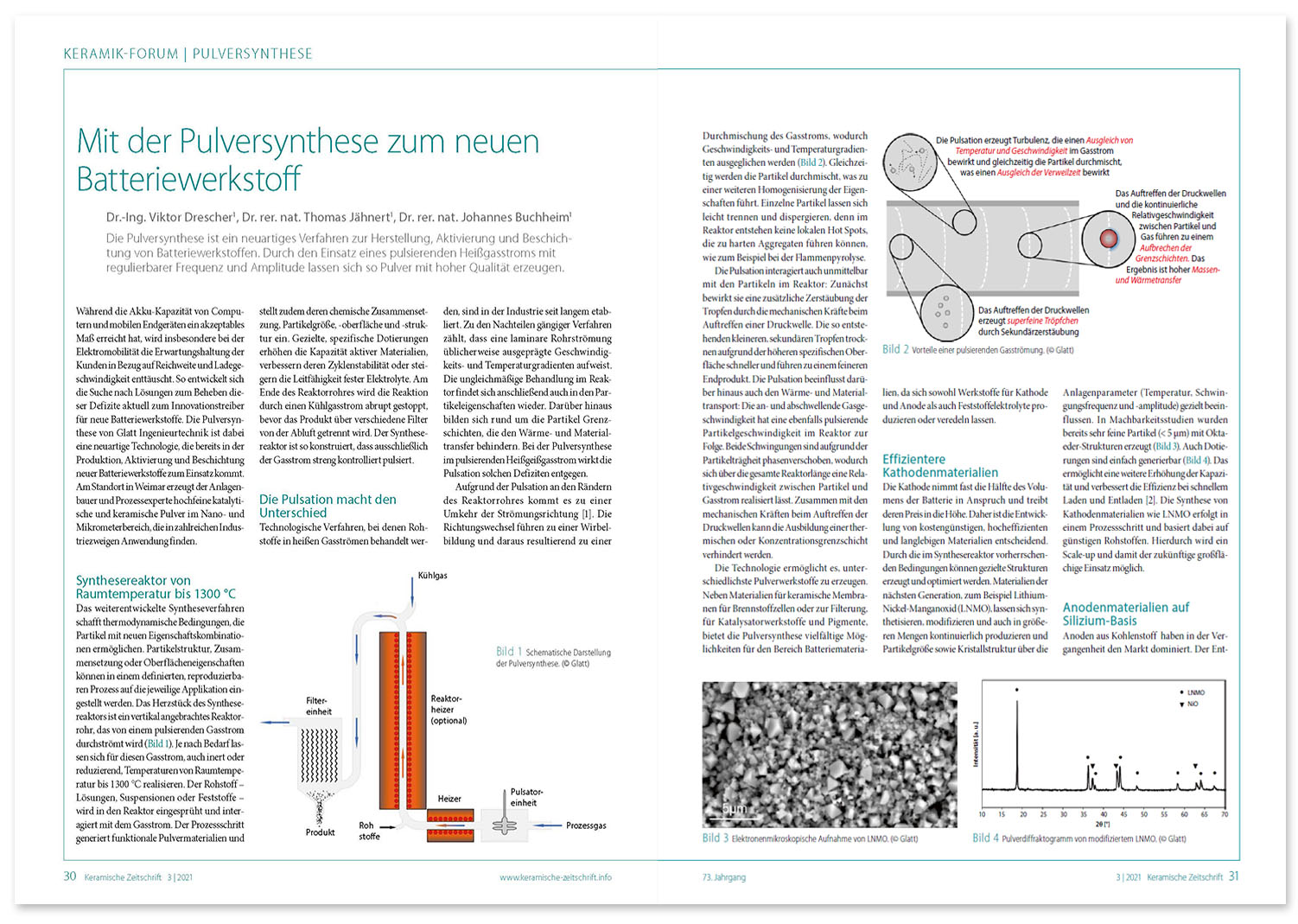
Veröffentlichter Fachbeitrag: ‚Mit Pulversynthese zum neuen Batteriewerkstoff‘, PDF, deutsch






 Copyright: publish-industry Verlag GmbH
Copyright: publish-industry Verlag GmbH